 SolutionsĪny copper(II) salt in solution will react with sodium hydroxide solution. If they boil at 100 and freeze at 0 at 1 atmosphere, then the sample is pure. To test whether the water sample is pure, the boiling and freezing points of the sample can be measured. This is because silver halides are insoluble in water. If you add silver nitrate solution to a sample of water containing halide ions, the silver halide is precipitated. A solution can be tested to see if it contains halide ions (chloride, bromide or iodide ions) by using silver nitrate. Halogens of chlorine, bromine or iodine have ions called halide ions. Testing for chlorides, bromides and iodides The acid reacts and gets rid of the carbonate ions. For example, if acid is not added, a white precipitate will be formed if there was a carbonate present, as barium carbonate is also white and insoluble. When acid is added to the solution, it destroys other compounds which might form white precipitates when barium chloride solution is added. This is insoluble in water and will be a white precipitate. Adding barium chloride solution to a sample of water containing sulphate ions will produce barium sulphate. You can see if a solution contains sulphate ions by using barium chloride. It also makes damp starch-iodide paper turn a blue-black. Chlorine makes damp blue litmus paper turn red which then turns white. Testing for chlorineĬhlorine has a lower density than air and it is collected downwards into a test tube or gas jar. Ammonia has a lower density than air and is very soluble in water. Testing for ammoniaĪmmonia makes damp red litmus paper blue and a white smoke of ammonium chloride is formed when hydrogen chloride gas from concentrated hydrochloric acid is held near it. Another test that can be used is to put bubbles of a gas through limewater – if the limewater turns cloudy then you know that carbon dioxide is present. Testing for carbon dioxideĪ lit splint will go out in a test tube full of carbon dioxide, however, this is also true for other gases, so this test does not ensure we have carbon dioxide specifically. If no oxygen is present, the splint will go out. This means that we can take a test tube of oxygen and place a glowing splint into this – the splint will relight in the presence of oxygen. To test for oxygen, we can use our knowledge of this element being flammable. A lit splint is placed at the top of a test tube of hydrogen and when a ‘squeaky pop’ noise is made, this indicates that hydrogen is reacting with oxygen in the air. Hydrogen reacts with oxygen when there is a flame or spark, to give water. Typically, hydrogen is composed of zinc and sulphuric acid with a bit of copper sulphate solution added to quicken the process. 
ExamplesĪ blue precipitate of copper(II) hydroxide is formed when sodium hydroxide solution is added to copper(II) sulphate solution. This may have been formed when a few drops of sodium hydroxide are added to a solution of a metal compound. 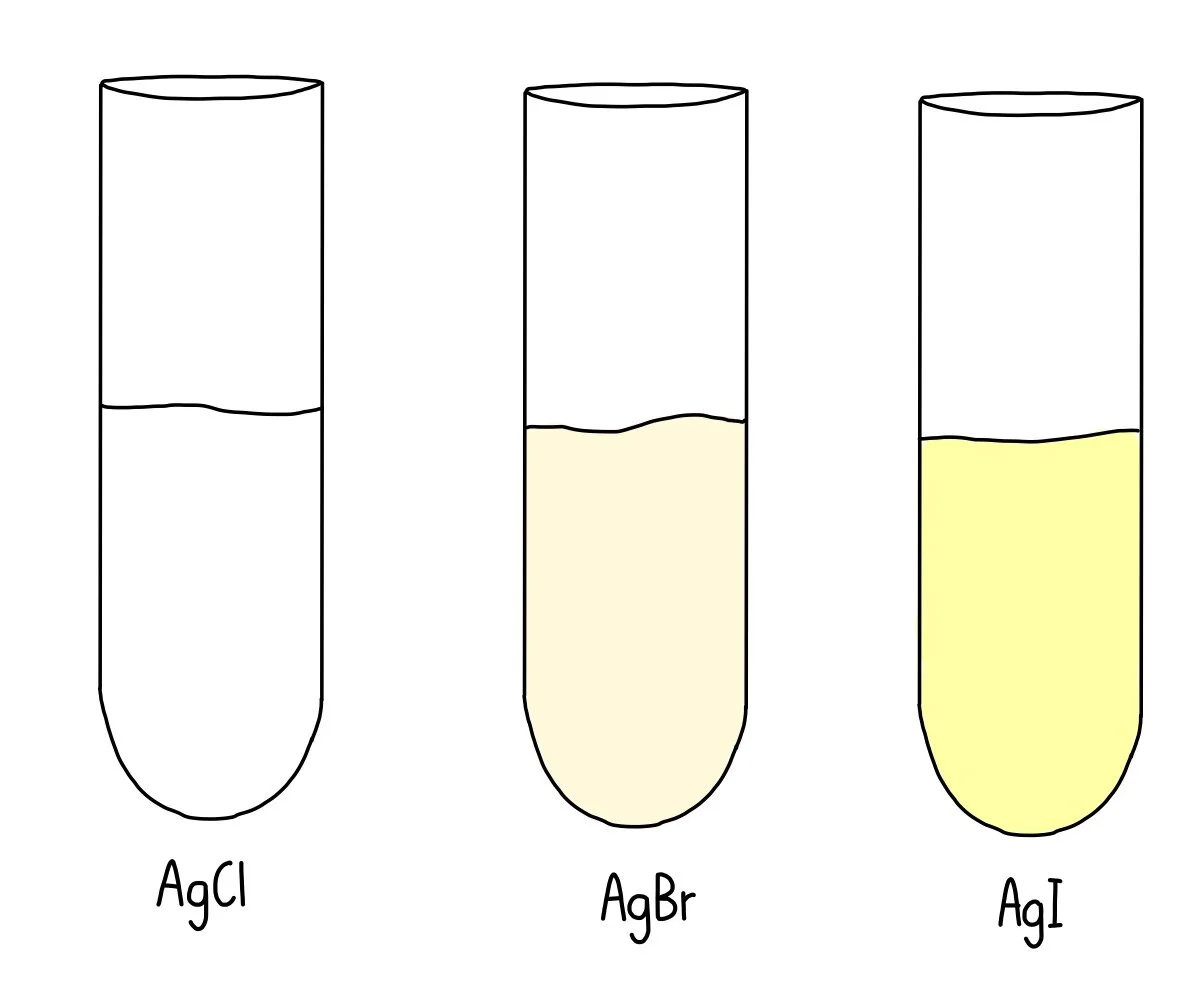
Precipitates come in the form of small particles that have been suspended in a solution. A precipitate is an insoluble solid that has been formed in the reaction. Identifying metal cations by using precipitation reactionsĪs most metal hydroxides are insoluble, they can be precipitated from aqueous solutions of metal salts by adding an aqueous solution of sodium hydroxide. The gas that’s been produced will turn damp red litmus paper blue. If ammonium ions are present, then a strong-smelling gas will be produced. To identify the ammonium ion, NH 4 +, add aqueous sodium hydroxide to the solid or solution, test and heat up the mixture. We can conduct experiments on positively charged ions (cations) to help us see which elements are present and to also examine the properties of these. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
